It is generally acknowledged that human mother’s milk is the best nutrition for newborn infants. Next to providing the energy and nutrients required for growth, it contains many bioactive molecules that contribute to the baby’s health. One very important group of its compounds, are human milk oligosaccharides (HMOs). These molecules, all consisting of a lactose core, are distinguished by their substitution with one or more neutral and/or acidic monosaccharides (figure below). Due to a plethora of modifications more than 200 unique structures can be detected. HMOs fulfil multiple beneficial roles. First of all, they have prebiotic properties, and are able to prevent the adhesion of certain pathogens and thus contribute to the lower incidence of intestinal, upper respiratory and urinary tract infections in breast-fed compared to formula-fed infants. Furthermore, HMOs are able to modulate epithelial cell responses in the intestines, and they lower the risk for necrotizing enterocolitis, an often fatal necrosis observed in preterm infants. Finally, HMOs provide the infant with essential nutrients for development of the brain, the intestinal tract and the immune system. Serving as antimicrobials, HMOs also have applications in elderly nutrition, pet and animal feed.
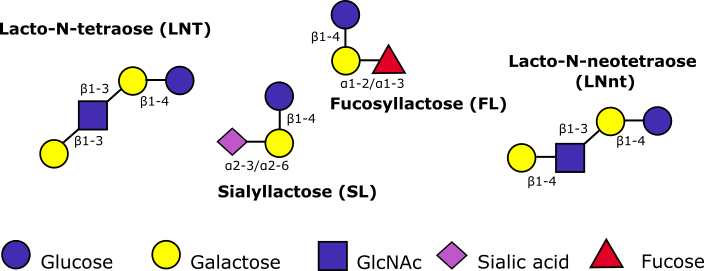
Some examples of human milk oligosaccharides. Abbreviations: GlcNAc, N-acetylglucosamine
Despite these proven and unique benefits, addition of human milk oligosaccharides to existing formulas is mostly impossible due to the lack of an industrial production method for these compounds. Currently available products do not contain any HMOs, but are fortified with HMO-like carbohydrates to mimic real mother’s milk as much as possible. Even though these additives possess prebiotic properties, none of them are able to fully substitute the natural compounds. Also considering the fact that 70% of the Belgian babies and 60% worldwide is formula-fed once they are 3 months old, the need for industrial production methods for HMOs is evident. Current production methods are based on extraction, chemical synthesis and biocatalysis. Unfortunately, it is nearly impossible to obtain the more complex compounds in a pure or in a sufficient amount with existing procedures. A more promising method to ensure a sufficient supply of HMOs is the production by means of microorganisms. Microorganisms use renewable resources and do not ask for extreme reaction conditions, which makes this production method more sustainable compared to existing production routes. In addition, extra attention to stereoselectivity is less required and cofactors are supplied by the host, making this way of working with nature extremely promising for the production of HMOs.
InBio.be started research on the microbial production of HMOs about 10 years ago. Integrated microbial host engineering and process design meanwhile led to the industrial scale production of fucosyllactose using Escherichia coli, a process which is now being commercialized at our spin-off company Inbiose. This proves that microbial production of HMOs is a promising route to follow, yet, there is still ample room for improvement. For example, industrial scale production of sialylated HMOs or more complex HMOs is not possible yet. Hence, we continue to improve our platform strains for the synthesis of these HMOs and we are striving to diversify the product portfolio. Advanced metabolic engineering and synthetic biology are combined with bioprocess development to turn these platform strains into modern, robust and versatile cell factories for HMO production.
For more information about HMOs and possible collaborations, please contact Prof. Wim Soetaert or Dr. Sofie De Maeseneire.
